Veterinary Medicines Directorate
476 articles
Showing 91-120
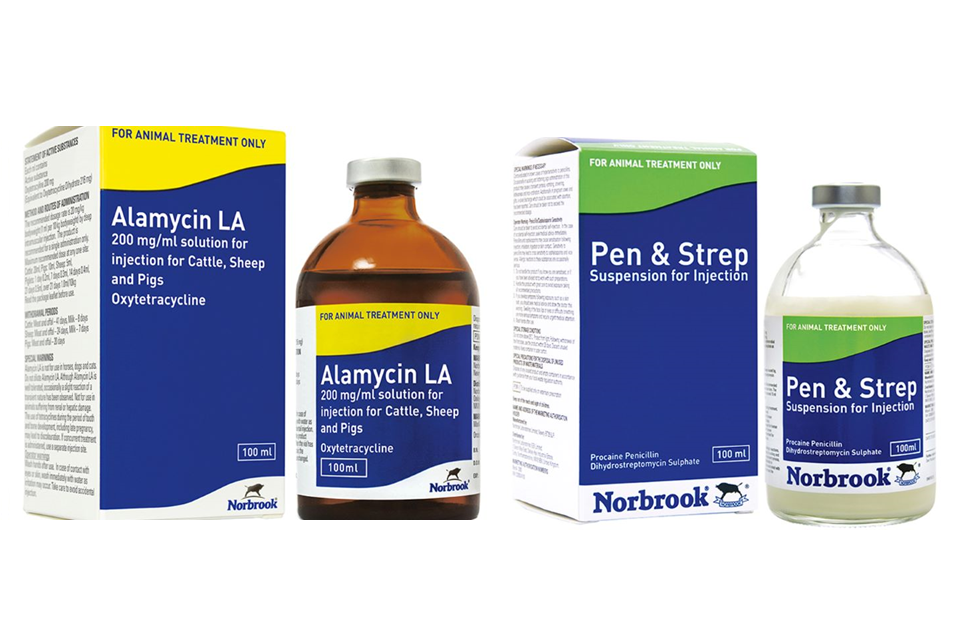
Product defect recall alert – Alamycin LA Injection, Pen + Strep Injection
Thursday, 05 June 2025
Recall alert for Alamycin LA Solution for Injection for Cattle, Sheep and Pigs 100ml and Pen & Strep Suspension for Injection 100ml. ...

Recruitment for a Distribution and Supply Chain Inspector for South East England
Thursday, 29 May 2025
Distribution and Supply Chain Inspector vacancy, working on inspections and investigations to ensure compliance with the Veterinary Medicines Regulati...

Recruitment for an IT Service and Operations Manager
Thursday, 29 May 2025
IT Service and Operations Manager, working on the running and maintenance of the VMD services available to users....

SPC change Credelio Chewable Tablets for Dogs
Tuesday, 29 April 2025
Change to the information provided on adverse events in the Summary of Product Characteristics for Credelio 56mg/ 112mg/ 225mg/ 450mg/ 900mg Chewable ...

NT Labs Methylene Blue Product defect recall alert
Tuesday, 08 April 2025
Product recall for two batches of NT Labs Methylene Blue....

Recruitment for Synergy Workstream Leads
Tuesday, 01 April 2025
Workstream Lead vacancy working on the Synergy Programme....
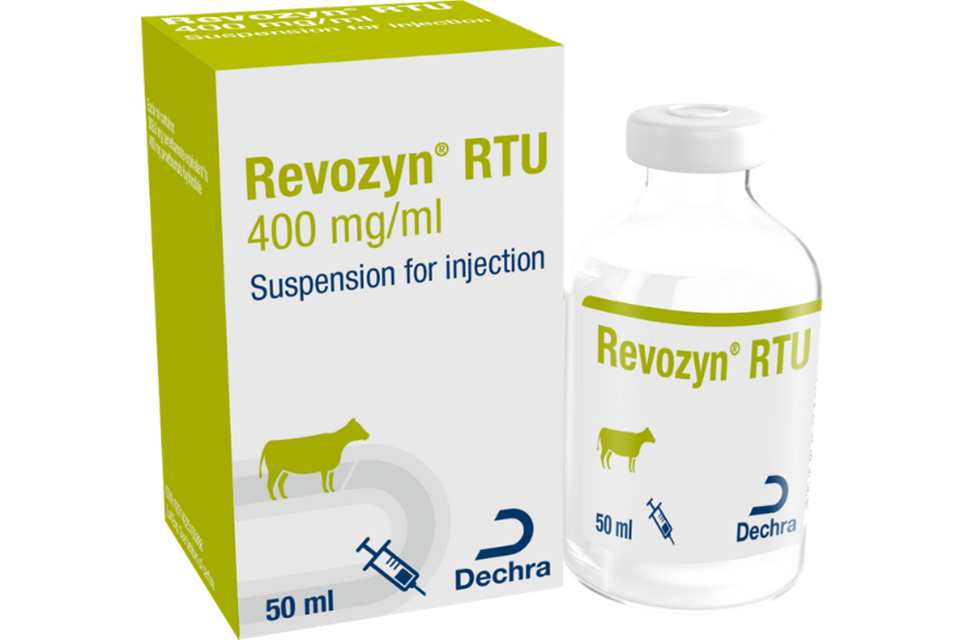
Revozyn RTU 400 mg/ml Suspension for Injection for Cattle Recall alert
Tuesday, 25 March 2025
Product defect recall alert for Revozyn RTU 400 mg/ml Suspension for Injection for Cattle by Dechra on behalf of Eurovet Animal Health B.V. ...

Alizin 30 mg/ml Solution for Injection – SPC change
Monday, 17 March 2025
Change to the information provided on adverse events in the Summary of Product Characteristics for Alizin 30 mg/ml Solution for Injection....

Update: Genta Equine 100 mg/ml Solution for Injection for Horses – Adverse events
Friday, 14 March 2025
More information following investigation into an increase of adverse event reports following the use of Genta-Equine 100 mg/ml Solution for Injection ...

Recruitment for a Cross Defra coordinator for the AMR National Action Plan
Wednesday, 12 March 2025
Cross-Defra Coordinator vacancy working for the Antimicrobial Resistance (AMR) National Action Plan (NAP)....

VICH consultation relating to stability testing for medicated premixes
Thursday, 06 March 2025
VICH consultation until 31 May 2025 on guideline relating to stability testing for medicated premixes....

Recruitment of a Non Executive Director for the Veterinary Medicines Directorate
Thursday, 06 March 2025
We are seeking a Non-Executive Director to join our Management Board and Chair the Audit and Risk Assurance Committee (ARAC). ...

Recruitment for Policy Officer
Wednesday, 26 February 2025
Policy Officers work on the development and implementation of policy and legislation on veterinary medicines....

Extramural Studies Placement 2025
Thursday, 13 February 2025
Opportunity for veterinary students to apply to attend a one-week extramural studies (EMS) placement in July 2025. ...

Recruitment for a Senior Safety Assessor
Friday, 31 January 2025
Senior Safety Assessor vacancy, working on authorising veterinary medicines in the UK....

Solensia 7 mg/ml Solution for Injection for Cats
Thursday, 30 January 2025
VMD response to concerns raised following media reports of serious adverse events in cats administered Solensia. ...

Recruitment for Application Co ordinators
Wednesday, 29 January 2025
Vacancies for Application Co-ordinator roles, working on applications received in respect of veterinary medicine marketing authorisations....

Recruitment for Higher Executive Officer in the Director Support Office
Thursday, 23 January 2025
Director Support Office vacancy, working to support our Directors and Senior Colleagues....

Training Event: Veterinary Dispensary Manager Online Course
Tuesday, 21 January 2025
Online training event on Wednesday 5 March 2025 ideal for anyone involved in veterinary dispensary roles....

Librela solution for injection in dogs
Wednesday, 15 January 2025
VMD response to concerns raised following media reports of serious adverse events in dogs administered Librela. ...

Meflosyl 5% Solution for Injection SPC change
Monday, 13 January 2025
Change to the information provided on adverse events in the Summary of Product Characteristics for Meflosyl 5% Solution for Injection, authorised for ...

VMD seizes over 13,000 unauthorised horse medicines sold illegally online
Wednesday, 18 December 2024
Over 13,000 AbPrazole and Absucralfate products destined for sites across the UK were seized by VMD enforcement officers. ...

Genta Equine 100 mg/ml Solution for Injection for Horses – Adverse events
Wednesday, 18 December 2024
Increase in suspected adverse event reports following use of Genta-Equine 100 mg/ml Solution for Injection for Horses....

Recruitment for Pharmacovigilance Assessors
Thursday, 12 December 2024
Pharmacovigilance Assessors vacancies within the Authorisations Division....

Recruitment for Head of Regulatory Affairs
Wednesday, 11 December 2024
Head of Regulatory Affairs vacancy, working on the authorisation, use, and manufacture of veterinary medicines in the UK....

Recruitment for an Antimicrobial Use, Stewardship and Stakeholder Engagement Manager
Wednesday, 11 December 2024
Antimicrobial Use, Stewardship and Stakeholder Engagement Manager vacancy, working on the VMD’s Antimicrobial Resistance (AMR) policy and surveillance...

Recruitment for a Senior External Communications Manager
Wednesday, 11 December 2024
Senior External Communications Manager vacancy, working on the VMD’s strategic communications....

Recruitment for a Synergy Delivery and Business Change Lead
Wednesday, 11 December 2024
Synergy Delivery and Business Change Lead vacancy, working on the delivery and implementation of the Synergy programme for the VMD....

Librela Solution for Injection for Dogs – SPC change
Tuesday, 10 December 2024
Change to the information provided on adverse events in the Summary of Product Characteristics for Librela Solution for Injection for Dogs (All streng...

Arthramid Vet – Adverse events
Sunday, 13 October 2024
Increased discussion of suspected adverse events in social media following use of Arthramid Vet in horses....